Ammonium formate

| |

| |
| |
| Names | |
|---|---|
| IUPAC name
Ammonium formate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| |
| |
| Properties | |
| CH5NO2 | |
| Molar mass | 63.056 g·mol−1 |
| Appearance | White monoclinic crystals, deliquescent |
| Odor | Slightly ammoniac |
| Density | 1.26 g/cm3[1] |
| Melting point | 116 °C (241 °F; 389 K) |
| Boiling point | 180 °C (356 °F; 453 K) decomposes[2] |
| (grams per 100g of water)102g(0 °C) 142.7 g (20 °C) 202.4 g (40 °C) 516 g (80 °C)[2] | |
| Solubility in other solvents | Soluble in liquid ammonia, ethanol, diethyl ether[2] |
| Thermochemistry | |
Std enthalpy of
formation (ΔfH⦵298) |
−556.18 kJ/mol |
| Hazards | |
| GHS labelling: | |
 [1] [1]
| |
| Warning | |
| H315, H319, H335[1] | |
| P261, P305+P351+P338[1] | |
| NFPA 704 (fire diamond) | <imagemap>
File:NFPA 704.svg|80px|alt=NFPA 704 four-colored diamond poly 150 150 300 300 150 450 0 300 Health 2: Intense or continued but not chronic exposure could cause temporary incapacitation or possible residual injury. E.g. chloroform poly 300 0 450 150 300 300 150 150 Flammability 1: Must be pre-heated before ignition can occur. Flash point over 93 °C (200 °F). E.g. canola oil poly 450 150 600 300 450 450 300 300 Instability 1: Normally stable, but can become unstable at elevated temperatures and pressures. E.g. calcium poly 300 300 450 450 300 600 150 450 Special hazards (white): no code desc none </imagemap> |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
410 mg/kg (mice, intravenous)[2] |
| Safety data sheet (SDS) | JT Baker MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Ammonium formate, NH4HCO2, is the ammonium salt of formic acid. It is a colorless, hygroscopic, crystalline solid.
Reductive amination
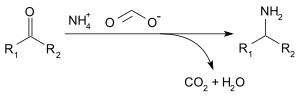
Acetone can be transformed into isopropylamine as follows:[citation needed]
- CH3C(O)CH3 + 2 HCO2− +NH4 → (CH3)2CHNHCHO + 2 H2O + NH3 + CO2
- (CH3)2CHNHCHO + H2O → (CH3)2CHNH2 + HCO2H
Uses
Pure ammonium formate decomposes into formamide and water when heated, and this is its primary use in industry. Formic acid can also be obtained by reacting ammonium formate with a dilute acid, and since ammonium formate is also produced from formic acid, it can serve as a way of storing formic acid.
Ammonium formate can also be used in palladium on carbon (Pd/C) reduction of functional groups. In the presence of Pd/C, ammonium formate decomposes to hydrogen, carbon dioxide, and ammonia. This hydrogen gas is adsorbed onto the surface of the palladium metal, where it can react with various functional groups. For example, alkenes can be reduced to alkanes, formaldehyde to methanol, and nitro compounds to amines.[3][4] Activated single bonds to heteroatoms can also be replaced by hydrogens (hydrogenolysis).
Ammonium formate can be used for reductive amination of aldehydes and ketones (Leuckart reaction), by the following reaction:[5]
Ammonium formate can be used as a mobile phase additive in high performance liquid chromatography (HPLC), and is suitable for use with liquid chromatography-mass spectrometry (LC/MS). The pKa values of formic acid and the ammonium ion are 3.8 and 9.2, respectively.
Reactions
When heated, ammonium formate eliminates water, forming formamide. Upon further heating, it forms hydrogen cyanide (HCN) and water. A side reaction of this is the decomposition of formamide to carbon monoxide (CO) and ammonia.
References
- ^ 1.0 1.1 1.2 1.3 Sigma-Aldrich Co., Ammonium formate. Retrieved on 2014-06-10.
- ^ 2.0 2.1 2.2 2.3 "Ammonium formate".
- ^ Ram, Siya; Ehrenkaufer, Richard E. (1984). "A general procedure for mild and rapid reduction of aliphatic and aromatic nitro compounds using ammonium formate as a catalytic hydrogen transfer agent". Tetrahedron Lett. 25 (32): 3415–3418. doi:10.1016/S0040-4039(01)91034-2. hdl:2027.42/25034.
- ^ Smith, Michael B.; March, Jerry (2007). March's Advanced Organic Chemistry (6th ed.). John Wiley & Sons. p. 1816. ISBN 978-0-471-72091-1.
- ^ Alexander, Elliot; Ruth Bowman Wildman (1948). "Studies on the Mechanism of the Leuckart Reaction". Journal of the American Chemical Society. 70 (3): 1187–1189. doi:10.1021/ja01183a091. PMID 18909189.
External links
![]() Media related to Ammonium formate at Wikimedia Commons
Media related to Ammonium formate at Wikimedia Commons