Thallium azide
| |
| Names | |
|---|---|
| IUPAC name
Thallium(I) azide
| |
| Other names
thallium azide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| TlN3 | |
| Molar mass | 246.40 g·mol−1 |
| Appearance | yellow-brown crystals |
| insoluble | |
| Structure | |
| Tetragonal, tI16[1] | |
| I4/mcm, No. 140 | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
very toxic |
| GHS labelling: | |
   
| |
| Danger | |
| NFPA 704 (fire diamond) | <imagemap>
File:NFPA 704.svg|80px|alt=NFPA 704 four-colored diamond poly 150 150 300 300 150 450 0 300 Health 4: Very short exposure could cause death or major residual injury. E.g. VX gas poly 300 0 450 150 300 300 150 150 Flammability 0: Will not burn. E.g. water poly 450 150 600 300 450 450 300 300 Instability 3: Capable of detonation or explosive decomposition but requires a strong initiating source, must be heated under confinement before initiation, reacts explosively with water, or will detonate if severely shocked. E.g. hydrogen peroxide poly 300 300 450 450 300 600 150 450 Special hazards (white): no code desc none </imagemap> |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Thallium azide, TlN3, is a yellow-brown crystalline solid poorly soluble in water. Although it is not nearly as sensitive to shock or friction as lead azide, it can easily be detonated by a flame or spark. It can be stored safely dry in a closed non-metallic container.
Preparation and structure
Thallium azide can be prepared treating an aqueous solution of thallium(I) sulfate with sodium azide. Thallium azide will precipitate; the yield can be maximized by cooling.
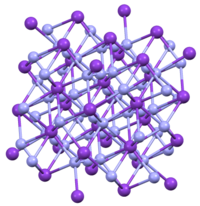
TlN3, KN3, RbN3, and CsN3 adopt the same structures. The azide is bound to eight cations in an eclipsed orientation. The cations are bound to eight terminal N centers.[2]

Safety
All thallium compounds are poisonous and should be handled with care; avoid breathing any dust or fumes.
References
- ^ Mauer F.A.; Hubbard C.R.; Hahn T.A. (1973). "Thermal expansion and low temperature phase transition of thallous azide". J. Chem. Phys. 59 (7): 3770–3776. Bibcode:1973JChPh..59.3770M. doi:10.1063/1.1680549.
- ^ Ulrich Müller "Verfeinerung der Kristallstrukturen von KN3, RbN3, CsN3 und TIN3" Zeitschrift für anorganische und allgemeine Chemie 1972, Volume 392, 159–166. doi:10.1002/zaac.19723920207